Kilimanjaro Summit
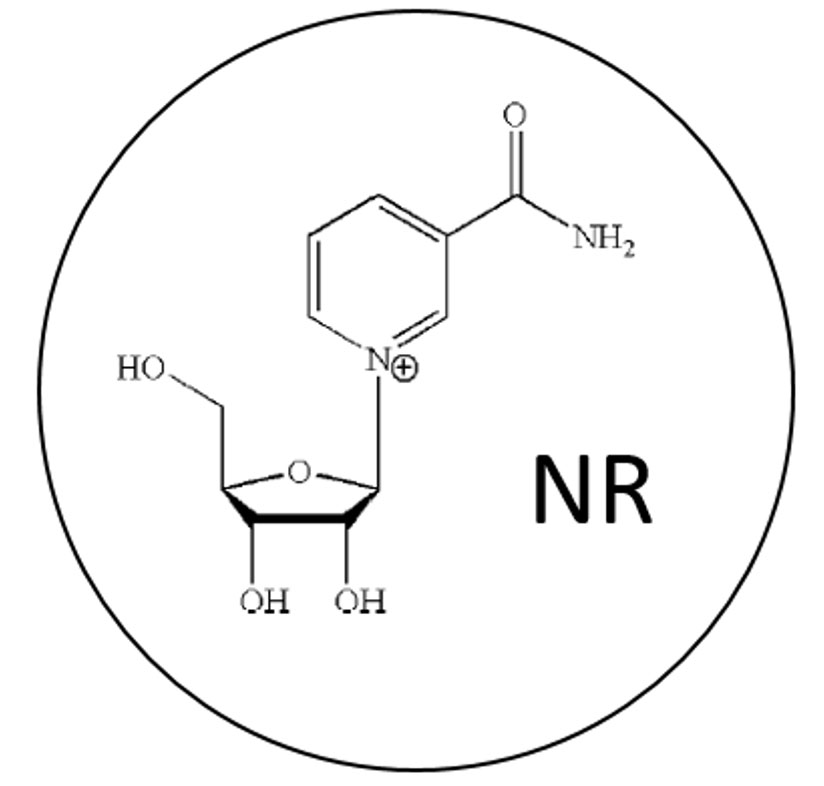
PURPOSE: To determine the effect of oral nicotinamide riboside (NR) on altitude tolerance, team cohesion, cardiopulmonary function, sleep, and measures of aging. This study also served as a Mars analogue mission, with further applications to military training and operations in hypobaric hypoxia field conditions.
LOCATION: Mount Kilimanjaro, Tanzania
DATE: August 2016
PRINCIPAL INVESTIGATORS:
Bruce Johnson, Ph.D., Mayo Clinic
Michael A Schmidt, Ph.D., Advanced Pattern Analysis & Countermeasures Group
EXPEDITION LEADER:
Measures
Test days:
- Demographic/ Medical History and Personality Questionnaires
- Echocardiogram
- Lung Function Testing
- Exhaled Nitric Oxide
- Bioimpedance
- Submaximal Exercise- Step test
- Cognitive Function
- Electrocardiogram (ECG), Photoplethysmography (PPG), respiratory belt and video camera assessed heart rates and respiratory rates (Phillips Vital Signs)
- Gut Microbiome (30 days prior to expedition, baseline in Africa and before departure home)
- Blood sampling
Daily:
- Altitude Adaptation Questionnaire
- Sleep Quality Assessment
- Monitoring Day and Night Time Pulse Oximetry
- Assessment of hydration
- Tracking Body Composition
- Tracking Energy Expenditure
- Team cohesion



